Lupus Accelerating Breakthroughs Consortium
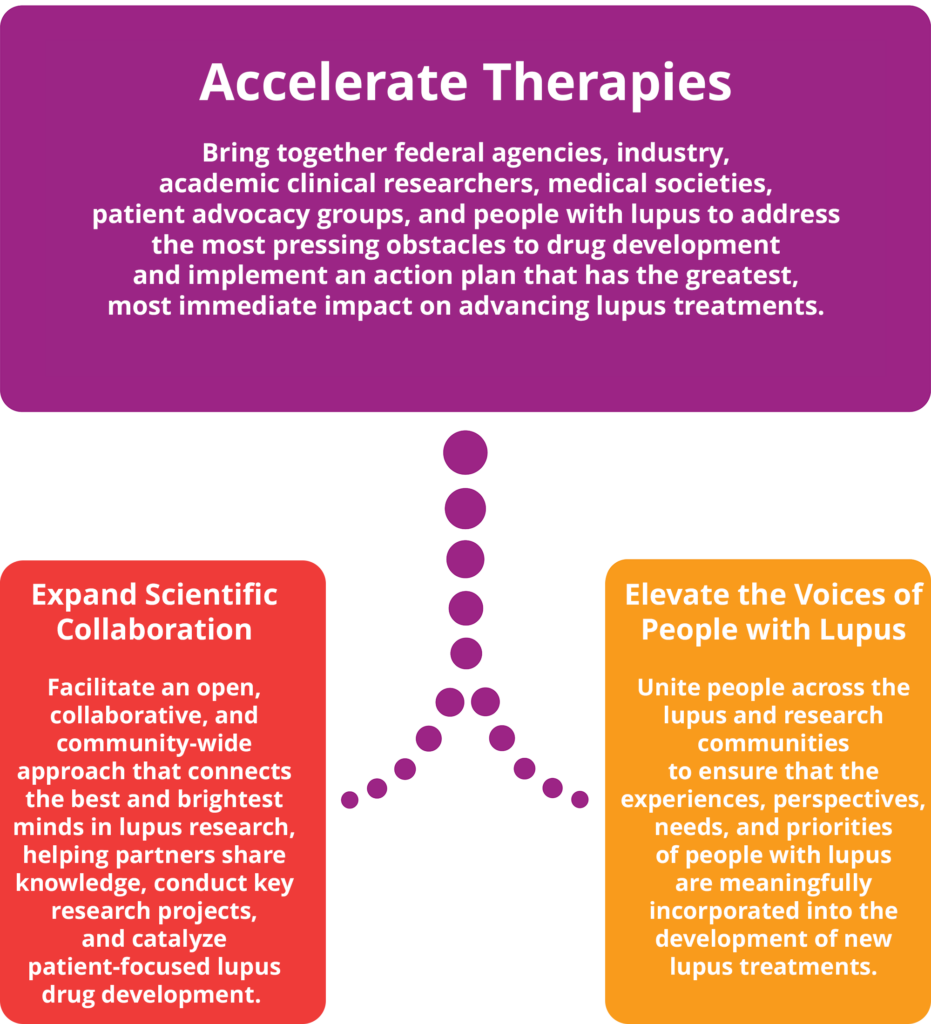
The Lupus Accelerating Breakthroughs Consortium (Lupus ABC) is the first public-private partnership with the U.S. Food and Drug Administration (FDA) that focuses specifically on advancing the development of safer and more effective treatments urgently needed for people with lupus. In collaboration with key stakeholders, Lupus ABC is bringing together people with lupus, federal agencies, medical societies, advocacy groups, industry, academic clinical researchers, and scientists, with the FDA, to overcome the scientific hurdles in drug development that are beyond the capacity of any single entity.
Lupus ABC will bring the concerns and needs of people living with lupus to the center of the drug development process, accelerating research to address their greatest unmet needs.
This is the Science of Us

Lupus impacts millions of people worldwide and over 300,000 people in the United States. This highly complex and potentially life-threatening autoimmune disease causes the immune system to produce antibodies that mistakenly recognize the body’s own cells as foreign, prompting other immune cells to attack and potentially damage organs such as the kidneys, brain, heart, lungs, blood, skin, and joints.
Lupus mainly affects women — 90% of people with lupus are female, and women are most often diagnosed between the ages of 15-44. Lupus is also up to three times more prevalent in women of color than white women. In the United States, lupus is the 5th and 6th leading cause of death among Black and Hispanic women ages 15–24 and 25-54, respectively — just behind cancer, heart disease, and HIV.
Lupus affects each person differently, presenting drastically varying symptoms among individuals, which makes the need for diverse treatment options all the more critical. Only four targeted therapies have been developed specifically to treat lupus in nearly 70 years.
Only four targeted therapies have been developed specifically to treat lupus in nearly 75 years.
Together we will change this.
Goals of Lupus Accelerating Breakthroughs Consortium